Tokyo, Japan, Mar 05: Scientists have uncovered important insights into how cells absorb and utilize externally supplied mitochondria—often referred to as the “powerhouses” of the cell potentially advancing the development of mitochondrial transplantation therapies aimed at restoring cellular energy function.
Mitochondria play a vital role in generating the energy required for cell survival, repair, and adaptation. In addition to energy production, they regulate cell death, calcium balance, and cellular responses to stress. When mitochondria malfunction, as seen in many neurodegenerative, inflammatory, and metabolic diseases, cells struggle to meet energy demands and maintain normal biological processes. To address this challenge, researchers are exploring mitochondrial transplantation, a novel therapeutic strategy that introduces healthy mitochondria into damaged or energy-deficient cells.
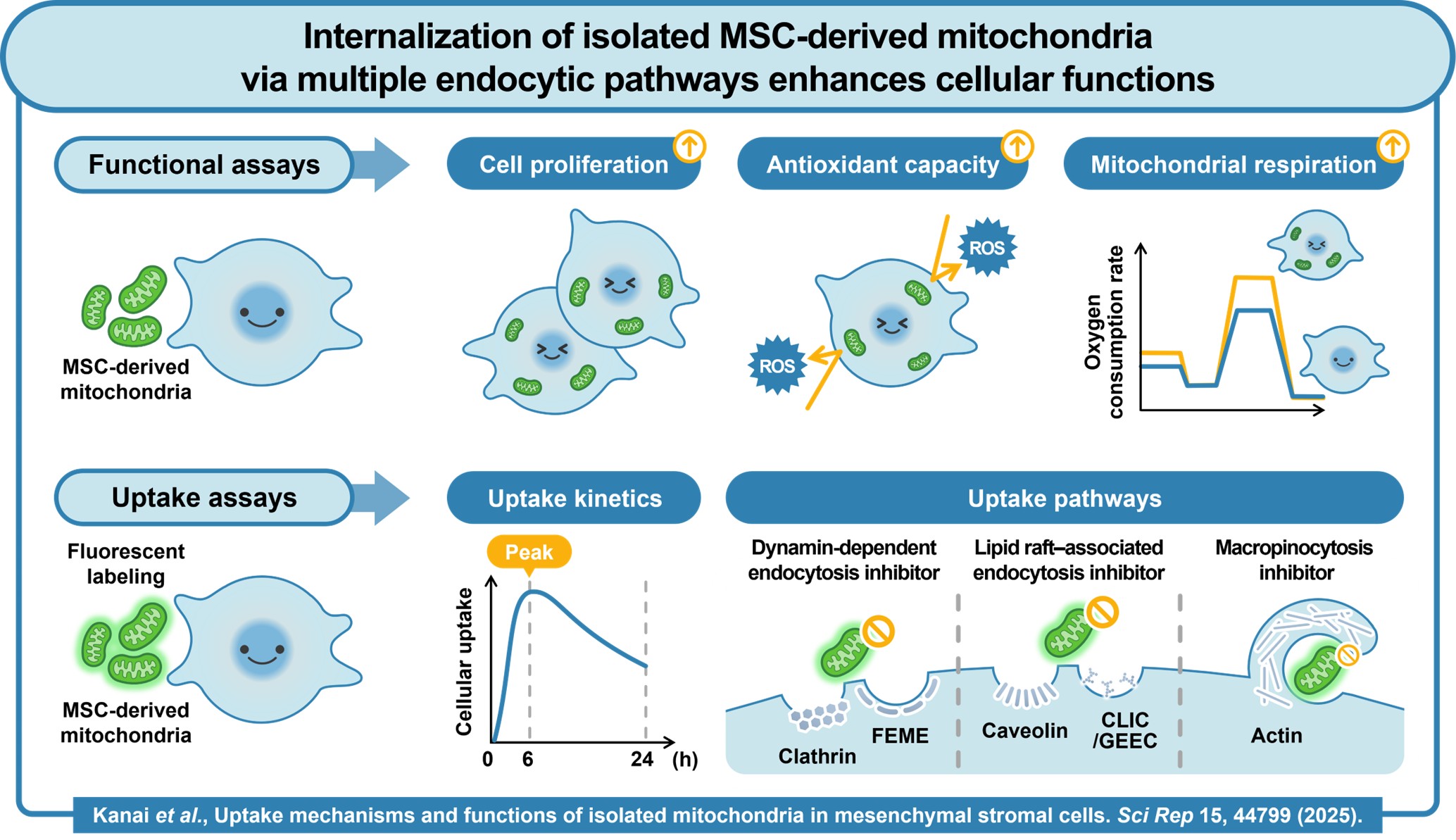
Despite promising findings from early experimental studies, a key challenge has slowed progress: scientists have not fully understood how transplanted mitochondria interact with recipient cells. Questions remained about whether mitochondria actively enter cells, how they cross the cell membrane, and whether they remain functional once inside.
A new study published in December 2025 in Scientific Reports by a research team led by Associate Professor Kosuke Kusamori from the Department of Life Sciences and Pharmaceutical Sciences at Tokyo University of Science (TUS) provides new answers. The study investigated how isolated mitochondria interact with mesenchymal stromal cells (MSCs)—cells widely used in regenerative medicine due to their therapeutic potential.
The research team isolated mitochondria from MSCs using a method designed to preserve their structural and functional integrity. Laboratory analysis confirmed that the isolated mitochondria retained their ability to produce adenosine triphosphate (ATP), the molecule responsible for cellular energy.
When these functional mitochondria were introduced to living cells, the researchers observed significant biological benefits. Cells treated with the mitochondria showed increased growth and demonstrated improved resistance to chemical and oxidative stress. Measurements of oxygen consumption—a key indicator of mitochondrial respiration—revealed enhanced cellular energy metabolism.
“Mitochondrial treatment increased the respiration rate, ATP production rate, and maximal respiratory capacity of MSCs in a concentration-dependent manner,” said Dr. Kusamori. “These findings suggest that isolated mitochondria preserve their intrinsic bioenergetic activity and can exert proliferative and cytoprotective effects on cells.”
To determine whether these benefits required mitochondria to enter the cells, the researchers used advanced imaging technologies, including fluorescence microscopy, confocal microscopy, flow cytometry, and live-cell imaging. The results showed that mitochondria were gradually internalized by cells over several hours. Electron microscopy further confirmed their presence inside membrane-bound vesicles within the cells.
Further experiments revealed that cells rely on multiple endocytic pathways—cellular processes used to absorb external substances—to internalize mitochondria, rather than a single dominant mechanism.
These findings provide critical insights into the biological mechanisms underlying mitochondrial uptake and function. Understanding these processes could help researchers improve the efficiency and safety of mitochondrial transplantation techniques in the future.
“This study provides a scientific foundation for developing therapies based on mitochondrial transplantation,” Dr. Kusamori explained. “It may pave the way for a new medical field called mitochondrial therapy, which directly restores cellular energy functions and supports safer and more sustainable treatments.”
Mitochondrial transplantation differs from stem cell or gene therapies because it does not alter a cell’s genetic identity. Instead, it restores energy production by supplying functional organelles, offering a potentially rapid solution for conditions involving mitochondrial dysfunction.
Researchers believe the approach could eventually help treat a range of disorders linked to impaired mitochondrial activity, including toxin-induced liver injury, ischemia-related conditions such as heart attack and stroke, and neurodegenerative diseases like Parkinson’s and Alzheimer’s.
However, scientists caution that mitochondrial transplantation remains in the preclinical stage. Further research is needed to confirm its long-term safety and effectiveness, optimize mitochondrial delivery methods, and ensure consistent quality and purity of transplanted mitochondria before clinical trials can begin.
Despite these challenges, the study represents a significant step forward in understanding mitochondrial biology and developing next-generation regenerative therapies aimed at restoring cellular energy and improving treatment outcomes for complex diseases.

